
Atlantic Cod in spawning aggregation. Photo: BluePlanetArchive / Wolfgang Poelzer
An understanding of the biology of aggregating fishes, information on the spatio-temporal distribution of spawning (and of spawning migrations), assessing the implications of fishing aggregations and determining best management interventions for exploited spawning aggregations (SAs) can be gained using scientific principles. Such understanding is key to safeguarding the function and persistence of SAs and the fisheries that they support. Increasingly, some aggregations are being appreciated and valued as the wildlife spectacles that they are. Aggregations are being documented in the SCRFA database.
Note that spawning gatherings (especially for non-tropical fishes) may not be referred to as ‘spawning aggregations’ but rather: spawning grounds, fishing grounds, fishing seasons, spawning runs, spawning habitats etc. The aggregation habit for spawning is widespread across many fish taxa, including many of high commercial importance, in many different aquatic ecosystems.
Studying and Managing Aggregations
A variety of methods, including a range of technologies, is now available to study and monitor SAs by both fishery-dependent and –independent means. In practice, methods and technologies should be adapted accordingly for each species and circumstance. Methods particularly applicable to reef fishes are summarized in the SCRFA Methods Manual. For a brief overview see here
General assessment categories are underwater visual census (UVC) surveys, which are particularly well-suited for shallow, clear, coral reef environments (fishery-independent : see below), and the use of catch data, including landings and related fisher information (fishery-dependent : see below). Other methods do not depend directly on adult fish presence (such as egg sampling or modelling). All categories can be supplemented, improved or substituted by various technologies, ranging from acoustic monitoring to various imaging systems.

Accurate measuring of fish length can be achieved using mounted cameras and stereo video while UVC surveys are being conducted. Photo: Mark Priest
Fishery-independent monitoring of spawning aggregations may be done using diver-based UVC, mostly in warmer, shallower waters, since aggregated fish can be observed and counted in-situ. However due to the dynamic nature of SAs, both in space and in time, and the often difficult field conditions where they occur, applying UVC often involves logistic and design challenges regarding accuracy/precision, temporal and geolocation elements. For example, an FSA may be too large or too deep to survey in entirety, or the fish are too numerous and move too quickly to count individually.

Diver ‘confronted’ with large aggregation. Photo: Mandy Eptison
In addition to in-water surveys, a range of other fishery-independent options are advancing aggregation research, for example to study migrating fish, assess population size, locate aggregations and determine fish numbers across diverse fish taxa. Methods range from video, still camera, GPS to track fishers and mark out sites, acoustics, tagging (internal and external), modelling (habitat, movement water conditions), egg sampling/tracking and genetics (e.g. recent illustrative references: Nanami et al., 2013; Lelievre et al., 2014; Rowell et al., 2015; Gonzalez-Irusta and Wright, 2016; Beldade et al., 2017; Erisman and Rowell, 2017; Zemeckis et al., 2019; Sherman et al., 2020; Waterhouse et al., 2020).

Tagging fish for FSA-related studies. Photo: Rick Nemeth
Fishery-dependent monitoring: Information on fish catches and fisher experiences are important for identifying the existence, condition and, sometimes, history of an aggregation, which are often not well documented over time. For example, short-lived peaks in high catches of ripe fish from a few locations are good indications that spawning aggregations are occurring.
However, assessing aggregation condition over time from fish catches alone can be problematic because the assumption that fish catch rates (usually measured as catch per unit of fishing effort, CPUE, such as catch per boat or per trap, etc.) reflect numbers of fish present in a linear fashion may not be valid.
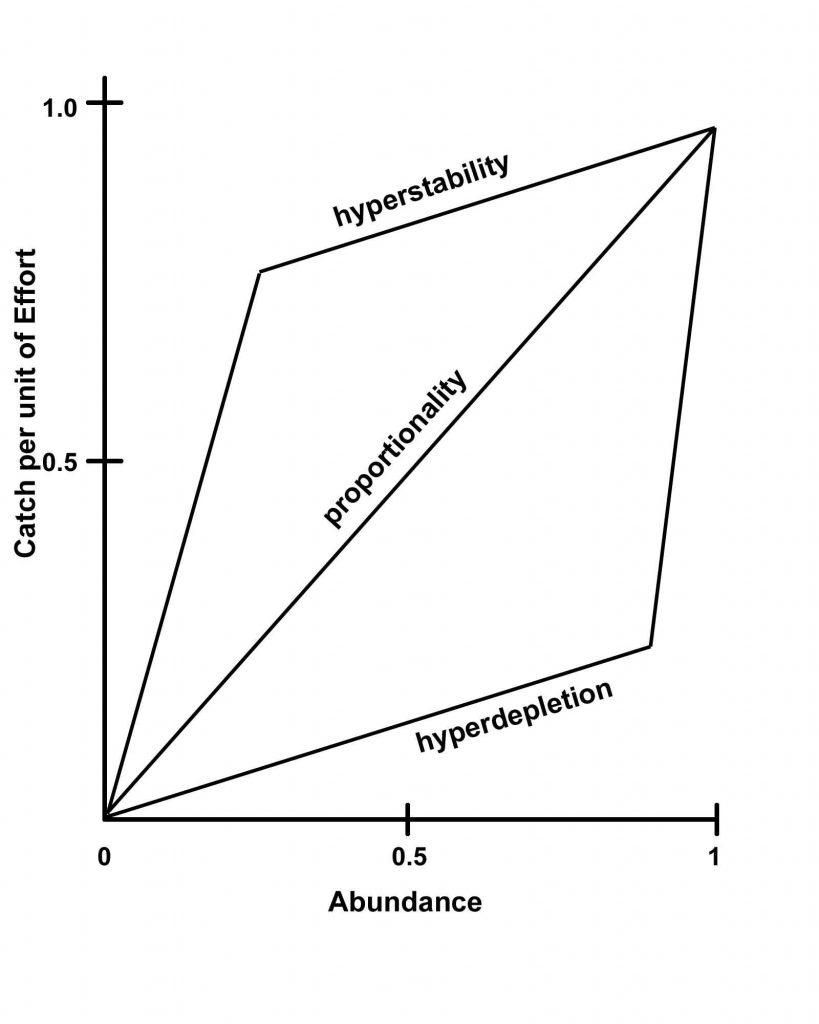
Hyperstability is when overall fish abundance and catch per unit effort are no long proportional and catch rates, therefore, no longer provide a good indication of fish abundance (which they usually are assumed to do in fisheries).
This is due to the phenomenon ‘hyperstability’, whereby CPUE becomes increasingly decoupled from true abundance and remains relatively stable as the underlying population declines and collapses (see graphic to the left) (illustrated via Natural Numbers Film). Hyperstability occurs because fish keep concentrating to spawn, even as their overall population numbers dwindle, because the imperative to reproduce is so strong. If fishing only occurs on its aggregations, fish catches may not obviously decline until the population becomes perilously low (this assumes that fishing is not so intense as to remove all fish aggregating at one time in one go-a possibility). By this late stage recovery may be challenging to achieve.
Interviews of, and collaborations with, fishers are important for understanding aggregation-fisheries, as well as the social and economic aspects of associated catches and acceptable options and implications of management. Studies that track fisher activities (e.g. Erisman et al. 2012), or use fisher knowledge and recall to understand SA fishing histories (e.g. Hamilton et al., 2012) are often the only means of documenting such aspects of SAs. Equity of access in fisheries of aggregating species is an important consideration only possible to determine through consultations with fisher communities.

Palauan fishers were interviewed about their catches over time and with different gears. Photos: Yvonne Sadovy
Managing Aggregations
Management of spawning aggregations includes one or a combination of measures according to the condition of the fishery, species, enforcement capability, fishing pressure, and social acceptance. Measures can involve both spatial and temporal components (spawning site/season closures), sales controls during the spawning season, minimum sizes, gear controls, etc. Sometimes aggregation protection may not be enough and other measures, such as minimum sizes or quotas year-round, are called for. Monitoring over time can determine the effectiveness of measures implemented. Absence of management for commercially exploited SAs almost invariably leads to aggregation losses or declines in these species and their fisheries.
Experience indicates that for commercially exploited spawning aggregations management with effective enforcement is almost always necessary to sustain associated fisheries long-term at their most productive levels. Depending on the species and circumstances, and with full and effective protection during aggregation seasons, it can take 5 to 10 years, or more, for recovery to begin or become evident (e.g. tropical groupers Nemeth, 2005; Hamilton et al., 2011; Waterhouse et al., 2020). Many temperate water aggregations have also declined and experience has shown that recovery may be difficult or prolonged if protection is implemented too little, too late, or if other matters are not considered such as the impact of fishing on spawning behavior (e.g. Oelofsen and Staby, 2005; Armstrong et al., 2013; Van Overzee and Rijnsdorp, 2014; Clarke et al. 2015).
Useful references for studying and managing aggregations:
Armstrong, M. P., Dean M. J., Hoffman, W. S., Zemeckis, D. R., et al. (2013). The application of small scale fishery closures to protect Atlantic cod spawning aggregations in the inshore Gulf of Maine. Fisheries Research 141: 62-69. https://www.sciencedirect.com/science/article/abs/pii/S0165783612002780
Beldade, Ricardo & Jackson, Alexis & Cudney-Bueno, Richard & Raimondi, Peter & Bernardi, Giacomo. (2014). Genetic structure among spawning aggregations of the gulf coney Hyporthodus acanthistius. Marine Ecology Progress Series. 499. 193-201. https://www.researchgate.net/publication/263388776_Genetic_structure_among_spawning_aggregations_of_the_gulf_coney_Hyporthodus_acanthistius
Clarke, J., Bailey, D. M., and Wright, P. J. (2015). Evaluating the effectiveness of a seasonal spawning area closure. ICES Journal of Marine Science, 72: 2627–2637.
https://academic.oup.com/icesjms/article/72/9/2627/2458713
Erisman, B., Aburto-Oropeza, O., Gonzalez-Abraham, C. et al. (2012). Spatio-temporal dynamics of a fish spawning aggregation and its fishery in the Gulf of California. Sci Rep 2, 284. https://doi.org/10.1038/srep00284. https://www.nature.com/articles/srep00284
Erisman, B. E., and Rowell, T. J., 2017. A sound worth saving: acoustic characteristics of a massive fish spawning aggregation. Biology Letters. https://doi.org/10.1098/rsbl.2017.0656
González-Irusta, J. M., and P. J. Wright. (2016). Spawning grounds of Atlantic cod (Gadus morhua) in the North Sea. ICES Journal of Marine Science, 73 (2):304–315. https://doi.org/10.1093/icesjms/fsv180
Hamilton, R. J., Potuku, T., and Montambault, J. R. (2011). Community‐based conservation results in the recovery of reef fish spawning aggregations in the Coral Triangle. Biol. Conserv. 144, 1850–1858.
http://www.conservationgateway.org/Documents/Hamilton,%20Potuku%20%20Montambault%202011.pdf
Hamilton, R.J., Giningele, M., Aswani, S., and Ecochard, J.L. (2012) Fishing in the dark – local knowledge, night spearfishing and spawning aggregations in the Western Solomon Islands. Biol. Conserv. 145, 246–257. https://www.researchgate.net/publication/256669040_Fishing_in_the_dark-local_knowledge_night_spearfishing_and_spawning_aggregations_in_the_Western_Solomon_Islands
Lelievre, S. Vazb, S. Martin, C. S., and Loots, C. (2014). Delineating recurrent fish spawning habitats in the North Sea. Journal of Sea Research, 91: 1-14 http://dx.doi.org/10.1016/j.seares.2014.03.008
Nanami A, Kawabata Y, Sato T, Yamaguchi T, Kawabe R, and Soyano K (2013) Spawning migration and returning behavior of white-streaked grouper Epinephelus ongus determined by acoustic telemetry. Marine Biology 161. doi:10.1007/s00227-013-2369-3
Nemeth, R.S. (2005). Population characteristics of a recovering US Virgin Islands red hind spawning aggregation following protection. Mar. Ecol. Prog. Ser. 286, 81–97. https://pubmed.ncbi.nlm.nih.gov/16612415/
Oelofsen, B., and Staby, A. (2005). The Namibian orange roughy fishery: Lessons learned for future management. Pages 555–559 in Shotton R, ed. Deep Sea 2003: Conference on the Governance and Management of DeepSea Fisheries, Queenstown, New Zealand, 1–5 December 2003. Part 1: Conference Reports, FAO Proceedings no. 3/1. FAO
Rowell, T., Nemeth, R., Schärer, M., and Appeldoorn, R. (2015). Fish sound production and acoustic telemetry reveal behaviors and spatial patterns associated with spawning aggregations of two Caribbean groupers. Mar. Ecol. Prog. Ser. 518, 239–254. doi: 10.3354/meps11060
Sherman, K. D., Paris J. R., King, R. A., Moore, K. A. , Dahlgren, C. P., Knowles, L. C., Stump, K. Tyler, C. R. and Stevens, J. R. (2020). RAD-Seq Analysis and in situ Monitoring of Nassau Grouper Reveal Fine-Scale Population Structure and Origins of Aggregating Fish Frontiers of Marine Research, 7 (157):15 pp. https://www.frontiersin.org/articles/10.3389/fmars.2020.00157/full
Van Overzee, H. M., and Rijnsdorp, A. D. (2014). Effects of fishing during the spawning period: Implications for sustainable management. Reviews in Fish Biology and Fisheries, 25: 1 –19. https://link.springer.com/article/10.1007/s11160-014-9370-x
Waterhouse,L., Heppell, S. A, Pattengill-Semmens, C. V., McCoye, C. Bush P., Johnson, B., and Semmens. B. X. (2020). Recovery of critically endangered Nassau grouper (Epinephelus striatus) in the Cayman Islands following targeted conservation actions. PNAS 117, 1587-1595. https://www.pnas.org/content/117/3/1587
Zemeckis, D. R., Dean, M. J., DeAngelis, A. I., Van Parijs, S. M., Hoffman, W. S., Baumgartner, M. F., Hatch L. T., Cadrin,S. X., and McGuire, C. H. (2019). Identifying the distribution of Atlantic cod spawning using multiple fixed and glider-mounted acoustic technologies. ICES Journal of Marine Science, Volume 76 (6):1610–1625, https://doi.org/10.1093/icesjms/fsz064.